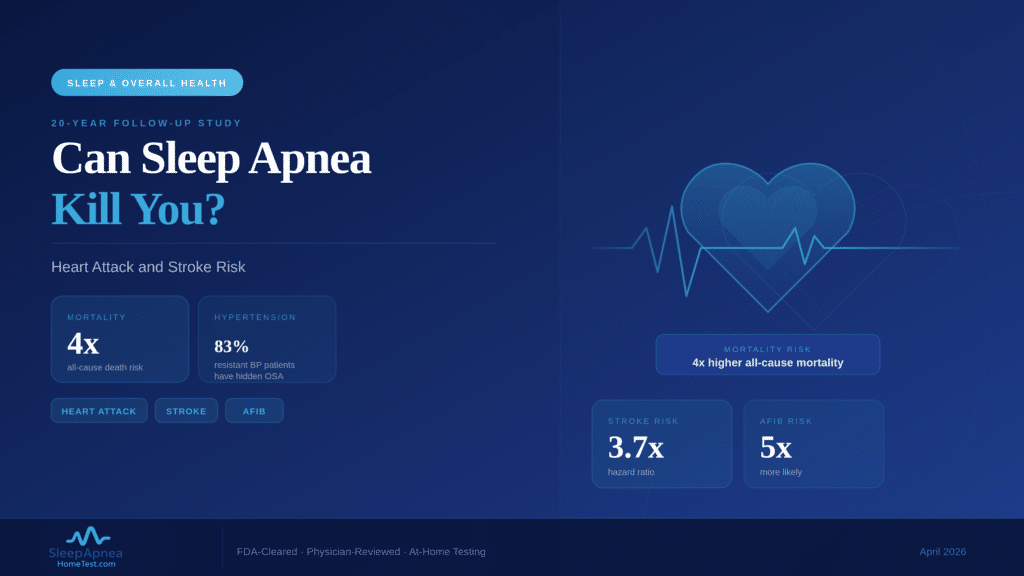
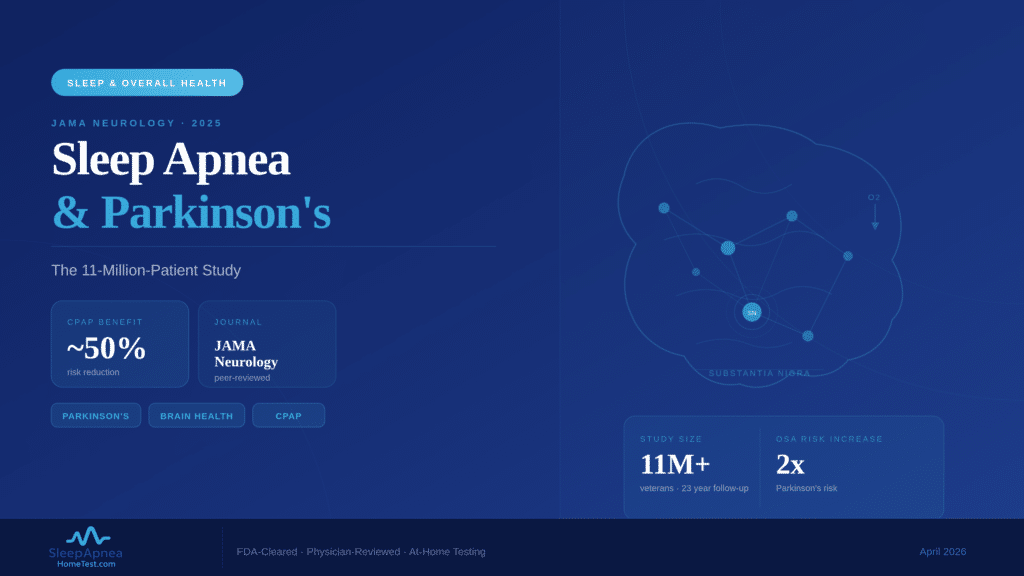
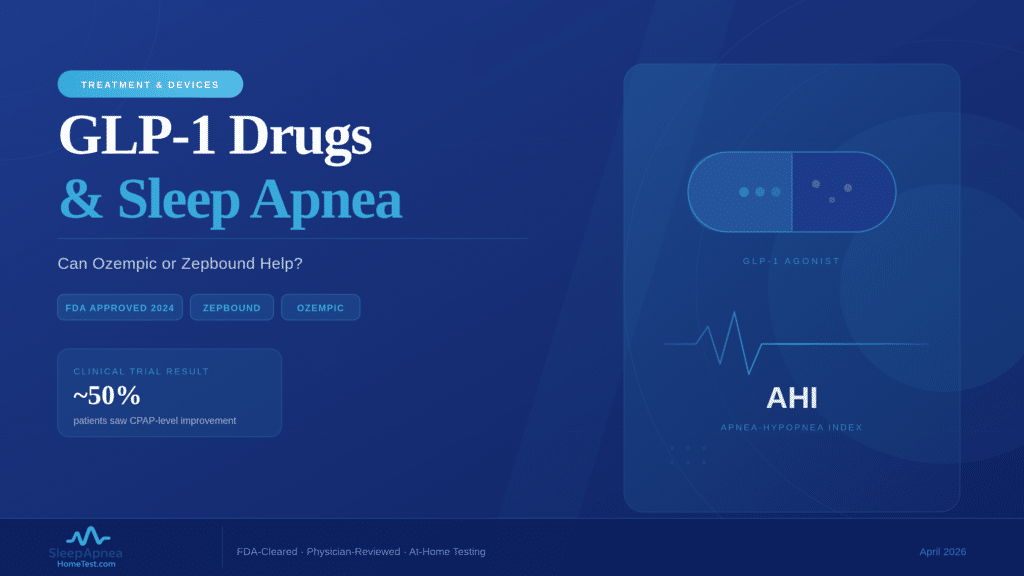
Zepbound (tirzepatide) became the first FDA-approved medication for obstructive sleep apnea on December 20, 2024, but it only applies to adults with moderate-to-severe OSA who also have a BMI of 30 or higher. Ozempic and Wegovy (both semaglutide) are not approved for sleep apnea, though some physicians prescribe them off-label when weight loss improves a patient’s breathing at night. Regardless of which GLP-1 drug you take or consider, a confirmed sleep apnea diagnosis through an FDA-cleared home sleep test is the required first step before any prescription.
What Are GLP-1 Drugs and How Do They Connect to Sleep Apnea?
GLP-1 receptor agonists mimic the glucagon-like peptide-1 hormone, which controls insulin secretion, slows stomach emptying, and reduces appetite. The result is significant weight loss: patients on semaglutide lose an average of 15% of body weight within 16 months, while those on tirzepatide lose 18-21% over 52 weeks. Because excess weight is the single strongest modifiable risk factor for obstructive sleep apnea, reducing body fat often shrinks the tissue pressing on the upper airway during sleep. A 2024 study in Sleep Medicine Reviews confirmed that a 10% increase in body weight raises OSA risk six-fold. The reverse also holds: dropping weight can reduce or, in some cases, resolve airway obstruction.
The Four Major GLP-1 Drugs: Which Ones Apply to OSA?
Four brand-name GLP-1 medications dominate the market in 2026. Each has distinct FDA approvals, and only one carries an OSA indication:
Zepbound (tirzepatide): The only GLP-1 drug FDA-approved for moderate-to-severe OSA in adults with a BMI of 30 or higher. A Phase 3 trial published in the New England Journal of Medicine (2024) found that tirzepatide reduced AHI by up to 63% over 52 weeks. Roughly half of trial participants improved enough to potentially discontinue CPAP therapy.
Mounjaro (tirzepatide): Same active ingredient as Zepbound but approved only for type 2 diabetes management. Not indicated for OSA or weight loss.
Wegovy (semaglutide): Approved for chronic weight management and cardiovascular risk reduction. Not approved for sleep apnea, but weight loss from Wegovy may reduce AHI scores in patients with obesity-related OSA.
Ozempic (semaglutide): Approved for type 2 diabetes. Not approved for sleep apnea. Some physicians prescribe it off-label when weight loss indirectly benefits OSA.
GLP-1 and Sleep Apnea Quick Facts
- FDA-approved for OSA: Only Zepbound (tirzepatide), since December 20, 2024
- Required for prescription: A confirmed AHI score from a diagnostic sleep study
- Average weight loss on tirzepatide: 18-21% of body weight over 52 weeks
- AHI reduction in trials: Up to 63% for tirzepatide vs. placebo
- Cost without insurance: Approximately $1,100/month for Zepbound
- Medicare coverage: Possible under Part D for OSA with obesity, requires prior authorization
- CPAP replacement potential: About 50% of trial participants improved enough to consider stopping CPAP
Why a Sleep Test Is Required for GLP-1 Prescriptions
A GLP-1 prescription for sleep apnea requires a documented diagnosis. Insurance companies, including Medicare Part D plans, demand a sleep study showing your Apnea-Hypopnea Index before approving prior authorization for Zepbound. Without an AHI score, no physician can prescribe tirzepatide for OSA, and no insurer will cover it. A home sleep apnea test provides this data. The test measures AHI, oxygen saturation (SpO2), snoring intensity, and airflow overnight. A board-certified sleep physician reviews the results and delivers a full diagnostic report, typically within days.
How Your Baseline AHI Guides Follow-Up Treatment
This baseline matters for tracking treatment effectiveness. Once you start a GLP-1 drug, your physician will order a follow-up sleep study after 6-12 months to measure whether your AHI dropped enough to adjust therapy. The initial test becomes the reference point for every future clinical decision. The testing requirement applies to off-label prescriptions too. If your physician prescribes Ozempic or Wegovy for weight loss and your sleep apnea improves as a side benefit, you still need a documented baseline and follow-up AHI before adjusting or discontinuing other OSA treatments like CPAP. Stopping therapy without data risks allowing residual apnea events to continue damaging your cardiovascular health overnight.
How GLP-1 Drugs Reduce Airway Obstruction During Sleep
The connection between weight loss and OSA improvement runs through three mechanisms. First, fat deposits around the pharynx (the upper throat) physically narrow the airway. Losing weight reduces this tissue, widening the passage air travels through during sleep. Second, excess abdominal fat pushes the diaphragm upward, reducing lung volume and lowering the “tug” that keeps the upper airway open. Weight loss reverses this mechanical disadvantage. Third, obesity drives systemic inflammation that swells airway tissues. GLP-1 drugs appear to reduce inflammatory markers, though researchers are still quantifying how much of the OSA benefit comes from anti-inflammatory effects versus pure weight reduction.
The 2024 SURMOUNT-OSA trial separated participants into two groups: those already using CPAP and those not using it. In both groups, tirzepatide produced significant AHI reductions compared to placebo, suggesting the drug works regardless of concurrent PAP therapy.
Insurance Coverage for GLP-1 Drugs and Sleep Apnea in 2026
Coverage depends on the drug, the diagnosis, and the plan. Zepbound may be covered under Medicare Part D when prescribed specifically for moderate-to-severe OSA in adults with obesity. The prescription must match the FDA-approved indication, and plans typically require prior authorization, a documented sleep study, and sometimes step therapy (trying CPAP first). Medicare does not cover any GLP-1 drug for weight loss alone.
Starting July 2026, the CMS “Medicare GLP-1 Bridge” demonstration program will provide early access to select GLP-1 medications for eligible beneficiaries, bridging to the broader BALANCE Model launching in 2027. This applies to drugs prescribed for covered indications such as OSA with obesity and cardiovascular risk reduction. Private insurers vary widely: some cover Zepbound for OSA with BMI documentation, while others exclude all GLP-1 drugs from formularies. Contact your plan directly with your ICD-10 code (G47.33 for OSA) and a copy of your sleep study results.
Can GLP-1 Drugs Replace CPAP? What the Evidence Says
Not yet, and likely not for everyone. The SURMOUNT-OSA trial showed approximately half of participants on tirzepatide improved to a level where CPAP discontinuation was clinically plausible. The other half still needed positive airway pressure therapy. Current clinical guidelines from the American Academy of Sleep Medicine (AASM) continue to recommend CPAP, APAP, or oral appliances as first-line treatments. GLP-1 drugs are positioned as adjunctive therapy, not a standalone replacement.
The bigger picture: OSA is not caused by weight alone. Jaw structure, tongue position, nasal anatomy, and neurological breathing control all contribute. A patient who is 30 pounds overweight with a recessed jaw may lose the 30 pounds on tirzepatide and still have significant apnea events. Only a diagnostic sleep test before and after treatment can determine whether the drug has resolved the breathing disorder.
What Happens if You Stop Taking the Drug?
Weight regain after discontinuing GLP-1 drugs is well-documented. Patients who stop semaglutide regain approximately two-thirds of lost weight within one year. If the weight loss was the primary reason for OSA improvement, the apnea events are likely to return as weight climbs back. This makes ongoing monitoring critical. Physicians managing OSA patients on GLP-1 drugs should schedule periodic sleep studies to catch any regression early, before the patient develops complications from untreated sleep-disordered breathing.
Side Effects and Risks of GLP-1 Drugs for Sleep Apnea Patients
GLP-1 medications carry side effects that sleep apnea patients should discuss with their physician before starting. The most common are gastrointestinal: nausea affects up to 44% of patients during the initial titration period, followed by vomiting, diarrhea, constipation, and abdominal discomfort. These symptoms typically decrease over 4-8 weeks as the body adjusts and the dose is slowly increased. Rare but serious risks include pancreatitis (approximately 0.2% incidence), gastroparesis (delayed stomach emptying that persists after stopping the drug), and potential thyroid concerns, though the thyroid risk has been observed primarily in animal studies.
For sleep apnea patients specifically, rapid weight loss raises a separate consideration. Losing muscle mass alongside fat can worsen respiratory function in some individuals. Protein-focused nutrition (1.6 grams per kilogram of body weight daily) combined with resistance training helps preserve lean muscle during GLP-1-assisted weight loss. This is especially relevant for OSA patients, because the muscles of the upper airway play a direct role in keeping the breathing passage open during sleep. Losing too much muscle without building it back can undermine the very improvement you are trying to achieve.
GLP-1 Drugs and Sleep Quality Beyond AHI
Weight loss from GLP-1 medications may improve sleep quality in ways that go beyond reducing apnea events. Participants in the SURMOUNT-OSA trial reported falling asleep faster, sleeping more soundly, and waking up feeling more rested. These improvements in subjective sleep quality appeared even before AHI reached normal levels, suggesting that reduced body weight improves sleep architecture (the structure of sleep stages) independently of airway obstruction.
Better sleep feeds a positive cycle. Adequate rest improves glucose regulation, reduces cortisol, and supports the willpower and energy needed for physical activity, all of which reinforce weight management. Sleep apnea treatment and GLP-1 therapy can work together as mutually reinforcing interventions. Identifying the apnea with a home sleep test sets the baseline. Treating it with CPAP or an oral appliance improves sleep immediately. Adding a GLP-1 drug (if clinically appropriate) addresses the weight component over months. Follow-up testing tracks progress and guides treatment adjustments.
What This Means for You
If you are overweight or obese and suspect sleep apnea, the path forward is straightforward: get tested first. A confirmed diagnosis unlocks treatment options including CPAP, oral appliances, and now GLP-1 medications like Zepbound. Without that diagnosis, neither your physician nor your insurer can move forward. The GLP-1 landscape is evolving rapidly, with new drugs entering trials and Medicare expanding access in 2026, but the starting point remains the same for every patient: a measured AHI score from an FDA-cleared sleep study that confirms whether obstructive sleep apnea is present and how severe it is.
Frequently Asked Questions
Can I get Zepbound prescribed for sleep apnea without a sleep study?
No. FDA approval specifies moderate-to-severe OSA in adults with BMI 30 or higher, and insurers require a documented AHI score before authorizing Zepbound for sleep apnea. An FDA-cleared home sleep test for $199 provides the AHI your physician needs for prior authorization.
Does Ozempic help with sleep apnea?
Ozempic is not FDA-approved for sleep apnea. It is approved for type 2 diabetes. Some physicians prescribe it off-label when weight loss is the goal, and weight reduction can improve OSA, but the drug has not been trialed specifically for AHI reduction. Only Zepbound (tirzepatide) has the OSA indication.
How much does Zepbound cost per month for sleep apnea?
Zepbound costs approximately $1,100 per month without insurance. Medicare Part D may cover it for moderate-to-severe OSA with obesity under prior authorization, and the CMS Medicare GLP-1 Bridge demonstration starting July 2026 expands access. Private insurance coverage varies. Contact your plan with ICD-10 code G47.33 and your sleep study results.
Can I stop CPAP if I start Zepbound?
Only with physician guidance and a follow-up sleep study. The SURMOUNT-OSA trial showed about half of participants on tirzepatide improved enough to potentially discontinue CPAP, but the other half still needed PAP therapy. Your AHI before and after treatment determines whether stopping CPAP is clinically safe. Get your baseline with a home sleep test.
What happens to my sleep apnea if I stop taking Zepbound?
Weight regain is well-documented after discontinuing GLP-1 drugs. Patients who stop semaglutide regain roughly two-thirds of lost weight within one year. If weight loss was the main driver of OSA improvement, the apnea events are likely to return as weight climbs back. Periodic follow-up sleep studies catch any regression before complications develop.
Next Steps
Your path to understanding whether a GLP-1 drug could help your sleep apnea starts with a diagnosis. Take the free 30-second AI facial scan to check your risk level. If your score is elevated, the home sleep test kit delivers the AHI data that physicians and insurers require. Your free consultation includes: a physician-reviewed diagnostic report, a personalized treatment recommendation, and guidance on insurance coverage for your next steps.
Medical disclaimer: This article is for educational purposes and does not constitute medical advice. Consult a licensed healthcare provider before starting, stopping, or changing any medication or treatment for sleep apnea.