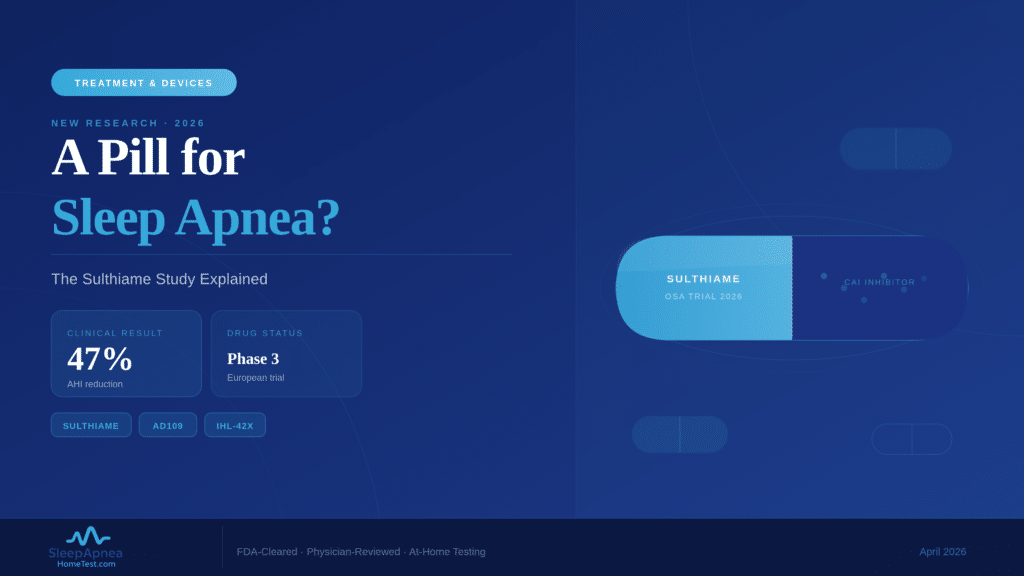
A European clinical trial published in March 2026 found that the drug sulthiame reduced breathing interruptions by up to 47% in adults with moderate-to-severe obstructive sleep apnea. Patients on the highest dose also showed improved blood oxygen levels during sleep. The study, led by the University of Gothenburg, is the most significant evidence to date that a pill could offer real relief for OSA, a condition currently treated primarily with CPAP machines, oral appliances, and surgery. Sulthiame is not yet FDA-approved for sleep apnea, and several years of regulatory review likely remain before it reaches pharmacy shelves, but the science is moving faster than many expected.
How Sulthiame Works: Stabilizing the Brain’s Breathing Signals
Sulthiame belongs to a class called carbonic anhydrase inhibitors. It was originally developed decades ago to treat epilepsy in children and has been prescribed for that purpose in parts of Europe. The drug’s mechanism for sleep apnea targets a different problem: unstable respiratory signaling in the brain. During sleep, the brainstem sends rhythmic signals that control the muscles keeping the upper airway open. In people with OSA, these signals can become erratic, especially during transitions between sleep stages. The airway muscles relax too much, the tongue falls back, and breathing stops.
Sulthiame stabilizes these signals by altering carbon dioxide sensitivity in the brainstem’s respiratory center. This keeps the neural drive to the airway muscles more consistent, reducing the likelihood of collapse. The approach is fundamentally different from CPAP therapy, which mechanically splints the airway open with pressurized air, and from GLP-1 drugs like Zepbound, which reduce OSA by producing weight loss.
OSA Medication Pipeline: 2026 Status
| Drug | Mechanism | AHI Reduction | US Status |
|---|---|---|---|
| Sulthiame | Stabilizes brain respiratory signaling (carbonic anhydrase inhibitor) | Up to 47% (2026 Gothenburg trial) | Not FDA-approved for OSA |
| AD109 (aroxybutynin + atomoxetine) | Increases upper airway muscle tone during sleep | Significant (2 Phase 3 trials, 2025) | FDA decision pending |
| IHL-42X (dronabinol + acetazolamide) | Dual: breathing stability + airway tone | Phase 2 data promising | FDA Fast Track (Dec 2025) |
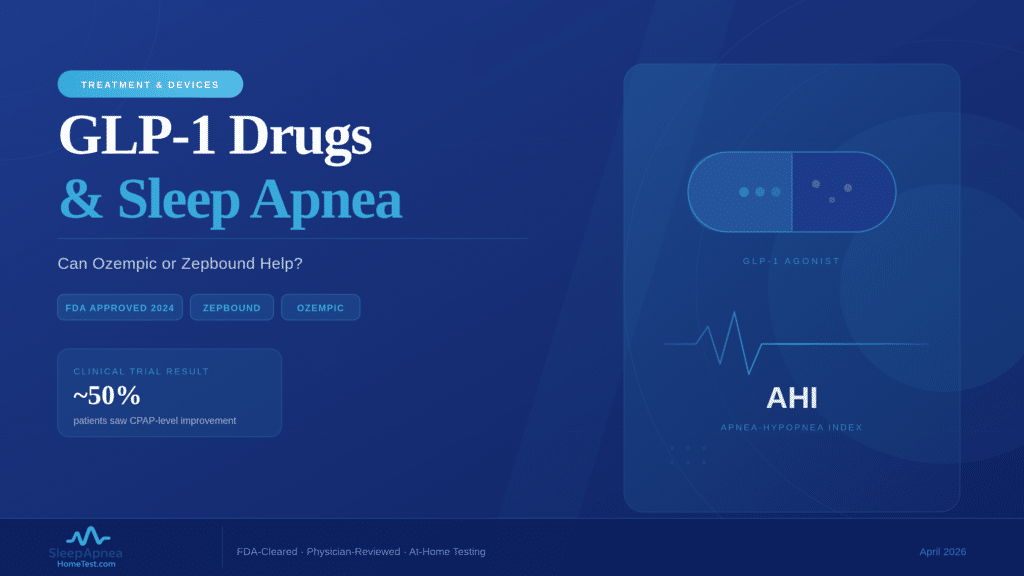
| Zepbound (tirzepatide) | Weight loss reduces pharyngeal fat | Up to 63% (SURMOUNT-OSA, 2024) | FDA-approved Dec 2024 (OSA + obesity) |
| Acetazolamide | Carbonic anhydrase inhibitor, stabilizes breathing control | ~38% (meta-analysis) | Off-label only |
| CPAP/APAP | Mechanically splints airway open | 70–80% with consistent use | Gold standard, established |
What the Trial Found: Numbers, Not Headlines
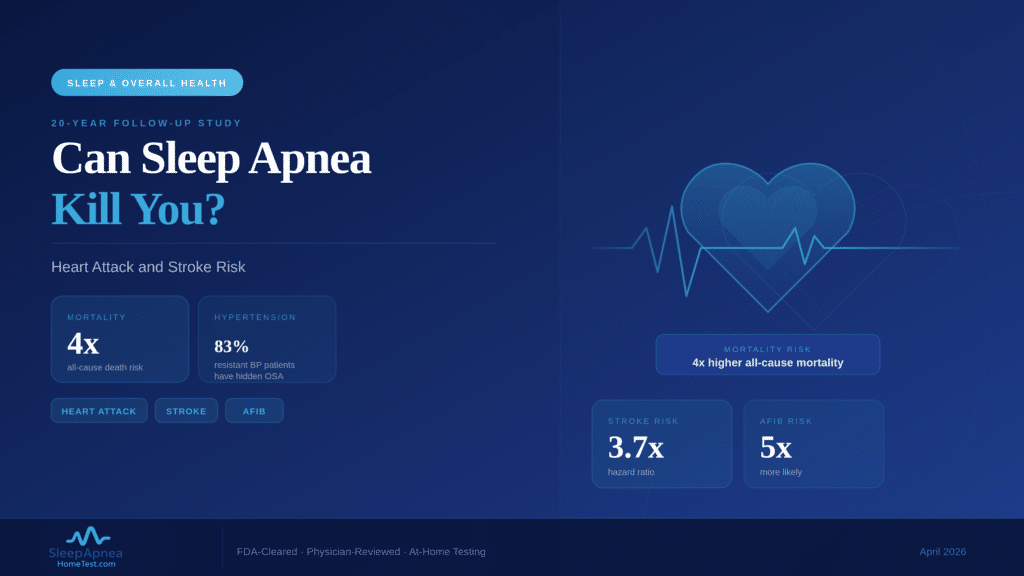
The Gothenburg trial enrolled adults diagnosed with moderate-to-severe OSA (AHI of 15 or higher). Participants were randomized to receive sulthiame at different doses or a placebo. After the treatment period, the highest-dose group showed a 47% reduction in their Apnea-Hypopnea Index. That means a patient averaging 30 breathing pauses per hour would drop to roughly 16. Oxygen desaturation events also decreased, and participants reported better subjective sleep quality. The lower-dose group showed smaller but measurable improvements, confirming a dose-dependent response.
To put this in context, consistent CPAP use typically reduces AHI by 70-80%, often bringing it below 5 (the threshold for normal). Sulthiame’s 47% reduction is clinically meaningful but does not match CPAP for patients with severe OSA. A patient starting at AHI 40 would drop to about 21, which still qualifies as moderate sleep apnea. This is why researchers describe the drug as a potential alternative for patients with milder presentations or as a supplement for those who cannot tolerate CPAP, not a wholesale replacement.
Sulthiame Safety Profile: What the Trial Reported
Sulthiame has a decades-long safety record in pediatric epilepsy, which gives researchers a baseline understanding of its side-effect profile. In the Gothenburg OSA trial, the drug was generally well tolerated. The most commonly reported side effects included tingling sensations in the hands and feet (paresthesia), which is a known effect of carbonic anhydrase inhibitors, and mild headache during the first weeks of treatment. Both tended to resolve as the body adjusted.
Carbonic anhydrase inhibitors can also affect electrolyte balance, particularly potassium and bicarbonate levels. Patients on sulthiame would likely require periodic blood tests to monitor these values, similar to monitoring protocols for acetazolamide. The trial did not report serious adverse events at rates higher than placebo, though the study period was relatively short. Longer-term safety data from extended trials will be needed before regulatory agencies grant approval for chronic nightly use in OSA patients. This is especially relevant because sleep apnea treatment is a lifelong commitment: unlike epilepsy, where medication may eventually be tapered, OSA patients would likely take the drug indefinitely unless the underlying causes (weight, anatomy) are addressed.
The Other Drugs in the Pipeline: AD109 and IHL-42X
Sulthiame is not the only medication advancing through clinical trials. Two other investigational drugs have produced significant results.
AD109 (Apnimed): This combination of aroxybutynin and atomoxetine works by increasing muscle tone in the upper airway during sleep. Atomoxetine is a norepinephrine reuptake inhibitor that activates the hypoglossal nerve, keeping tongue and throat muscles engaged. Aroxybutynin reduces REM-related muscle atonia. Two Phase 3 trials completed in 2025 showed significant AHI reductions over 26 weeks with favorable safety profiles. An FDA submission is expected, though no timeline has been confirmed.
IHL-42X (Incannex): This combination pairs dronabinol (a synthetic cannabinoid) with acetazolamide (a carbonic anhydrase inhibitor). The dual mechanism targets both breathing stability and airway tone. The FDA granted Fast Track designation in December 2025, which accelerates the review process for drugs addressing serious conditions with unmet medical need.
These drugs represent a shift toward “endotype-driven” treatment: matching the medication to the specific biological cause of a patient’s OSA, whether that is unstable breathing control, poor muscle tone, a low arousal threshold, or anatomical narrowing.
Why “Endotype-Driven” Treatment Matters
OSA is not a single disease. Four distinct biological mechanisms cause the airway to collapse, and most patients have a combination of them:
Anatomical narrowing: A physically small or crowded airway due to jaw structure, tongue position, enlarged tonsils, or fat deposits around the pharynx.
Poor muscle responsiveness: The throat muscles that keep the airway open during waking hours relax too much during sleep, especially during REM phases.
Unstable breathing control (high loop gain): The brain’s respiratory center overreacts to small changes in carbon dioxide, creating a cycle of over-breathing and under-breathing that triggers airway collapse.
Low arousal threshold: The brain wakes the patient at the slightest breathing difficulty, fragmenting sleep even when airway obstruction is mild.
CPAP addresses all four endotypes simultaneously by physically holding the airway open. A pill can only target one or two. That is why researchers expect the future of OSA pharmacology to involve matching the right drug to the right patient based on their specific endotype profile, not prescribing a single medication for everyone.
How Future Sleep Apnea Tests May Guide Drug Selection
The shift toward endotype-driven treatment has implications for how sleep apnea is diagnosed. Standard home sleep tests and polysomnography measure AHI, oxygen saturation, and airflow. These metrics confirm OSA and quantify severity but do not reveal which of the four endotypes is driving a patient’s specific condition. Newer research tools can measure loop gain (breathing control stability), arousal threshold, pharyngeal collapsibility, and muscle responsiveness. As these measurements become available outside research settings, physicians will be able to match each patient to the drug most likely to work for them.
For example, a patient with high loop gain (unstable breathing control) and normal airway anatomy might respond well to sulthiame or acetazolamide, which stabilize respiratory signaling. A patient with poor pharyngeal muscle tone during REM sleep might benefit from AD109’s muscle-activating mechanism. A patient whose primary driver is excess weight pressing on the airway might be best served by Zepbound or another GLP-1 drug. The common thread: every treatment path begins with a diagnostic sleep study that confirms OSA is present and measures the baseline AHI.
The CPAP Adherence Problem These Drugs Aim to Solve
CPAP therapy reduces AHI by 70-80% when used consistently, making it the most effective single treatment for OSA. The challenge is adherence. Approximately 50% of patients prescribed CPAP do not use it consistently, and many abandon it within the first year. Common complaints include mask discomfort, air leaks, nasal dryness, claustrophobia, and noise. For patients who have tried and failed CPAP, the promise of a pill is clear: swallow a tablet before bed, no mask, no machine, no noise.
Where Pills and CPAP May Work Together
The clinical picture is more nuanced. Sulthiame’s 47% AHI reduction at the highest dose is significant, yet it leaves residual apnea events in many patients. A patient starting at AHI 30 might drop to AHI 16, still in the moderate range. For this group, a pill may work best as part of a combined approach: medication to reduce severity, paired with a lower-pressure CPAP or an oral appliance to address the remaining events. Combination therapy could also improve CPAP adherence by reducing the pressure needed, making the mask more comfortable. For patients with mild OSA (AHI 5-14), a pill alone may be sufficient to bring the AHI below clinically significant levels, potentially sparing millions of patients from ever needing a CPAP machine.
What Still Needs to Happen Before a Pill Reaches Your Pharmacy
Several steps separate a successful clinical trial from a pill you can fill at your local pharmacy. Sulthiame needs regulatory submission to the FDA (or an expanded trial on U.S. soil), followed by a review period that typically runs 10-12 months for standard applications. AD109 and IHL-42X are further along in the U.S. regulatory pipeline but still await final decisions. Even after approval, insurance coverage determinations, prescribing guidelines, and physician education take additional time. A realistic timeline for the first non-weight-loss OSA pill reaching U.S. patients is 2027-2028, assuming no regulatory delays.
In the meantime, CPAP, APAP, oral appliances, positional therapy, and weight management remain the proven treatments. And every single one of these therapies starts with the same requirement: a confirmed sleep apnea diagnosis.
What This Means for You Today
The drug pipeline is encouraging, but it does not change the immediate action step for anyone who suspects sleep apnea. Whether you eventually treat your OSA with CPAP, an oral appliance, a GLP-1 drug, or a future pill like sulthiame, the process begins with a confirmed AHI score. That number determines your severity level, dictates which treatments you qualify for, and serves as the baseline for measuring improvement. A home sleep apnea test is the fastest, most accessible way to get that number.
Frequently Asked Questions
Is sulthiame available as a sleep apnea treatment in the US?
No. Sulthiame is not FDA-approved for sleep apnea in the United States. The 2026 Gothenburg trial was a European study, and US availability will require either FDA submission or a parallel US trial followed by a standard 10-12 month review. A realistic US timeline is 2027-2028 at the earliest.
How does sulthiame compare to CPAP for sleep apnea?
Sulthiame reduced AHI by 47% at the highest dose in the Gothenburg trial. CPAP typically reduces AHI by 70-80% with consistent use. For severe OSA, CPAP remains more effective. Sulthiame may work best for mild OSA alone, or combined with CPAP or an oral appliance for patients who cannot tolerate higher pressures.
What side effects does sulthiame have?
The most common side effects in the OSA trial were tingling in the hands and feet (paresthesia) and mild headache during the first weeks, both of which resolved as patients adjusted. Carbonic anhydrase inhibitors can affect potassium and bicarbonate levels, so periodic blood tests would likely be required during long-term use.
What other sleep apnea pills are in development?
Two other drugs are advancing: AD109 (aroxybutynin + atomoxetine) from Apnimed, which completed two positive Phase 3 trials in 2025 and targets airway muscle tone; and IHL-42X (dronabinol + acetazolamide) from Incannex, which received FDA Fast Track designation in December 2025. Both are further along in the US regulatory pipeline than sulthiame.
Will I still need a sleep study if a pill gets approved?
Yes. Every approved OSA treatment, including CPAP, oral appliances, Zepbound, and any future pill, requires a confirmed AHI score from a diagnostic sleep study. The AHI determines severity, qualifies you for insurance coverage, and serves as the baseline for measuring whether treatment is working. An FDA-cleared home sleep test gives you that number for $199.
Next Steps
The science of sleep apnea medication is accelerating, but your diagnosis is the foundation for any treatment path, now or in the future. Take the free AI facial scan to assess your risk in 30 seconds. If your score is elevated, the home sleep test kit delivers physician-reviewed results with an AHI score that qualifies you for every available treatment. Your test includes: an FDA-cleared monitoring device, overnight data collection in your own bed, and a board-certified sleep physician’s diagnostic report.
Medical disclaimer: This article is for educational purposes and does not constitute medical advice. Consult a licensed healthcare provider before starting, stopping, or changing any medication or treatment for sleep apnea.